The Bulletin of the United States Fish Commission 1901.
Page 103
Contributions from the Biological Laboratory of the U. S. Fish Commission, Woods Hole, Massachusetts.
The Reactions Of Copepods To Various Stimuli And The Bearing
Of This On Daily Depth-Migrations.
By G.H. Parker,
Assistant Professor of Zoology, Harvard University.
INTRODUCTION.
Although the marine copepods are almost microscopic crustaceans, they are so numerous and so widely distributed that they form one of the most important constituents in the surface life of the ocean. As a food for fishes, especially the newly hatched fry, they have been justly regarded by fish-culturists as almost indispensable, and their remarkable daily migrations have been a matter of growing interest and speculation for the naturalist.
From the standpoint of oceanic food supply, the copepods form an important link between the microscopic plants and the larger animals. In the ocean, as on the land, animals feed either directly on plants or on other animals that in turn feed on plants, and, though this regression may sometimes go back several steps, all animals are ultimately dependent on vegetable life for their food. Since plants are the only forms that have the capacity of elaborating food from strictly inorganic materials, they are in reality tne only independent organisms.
The vegetation of the ocean, except on the very shore, differs from that of the land chiefly in being composed almost entirely of plants of microscopic size. A vegetation of this kind can not give support to large herbivorous animal such as find easy nourishment in the luxuriant plant growth of the land, but it affords an abundant supply of food for small animals like the copepods. Many copepods feed almost exclusively on the minute plants of the sea, and then fall prey to small fishes, which in turn are eaten by larger ones. Thus the copepods form a link in the chain of food supply that reaches from the independent microscopic plants to the largest marine animals. Their place in this chain is admirably illustrated by Peck (1896, p. 353) in his account of the food of the squeteague.
In the stomach of a large squeteague Peck found an adult herring in which were two young scup besides many small crustaceans. The stomach of the scup contained copepods, and the alimentary canals of these showed remains of microscopic plants (diatoms). Thus plant substance served as food for copepods, copepods for scup, scup for herring, and herring for squeteague. This sequence makes evident the importance of copepods as collectors of vegetable food which after assimilation serves as nourishment for the larger fishes.
-----------------------------------------------------------
BULLETIN OF THE UNITED STATES FISH COMMISSION 1901.
Page 104
Although many copepods have been obtained from great depths in the ocean, they are, for the most part, surface or pelagic animals. This even applies to shallow waters. Thus, in Buzzards Bay, which is probably nowhere over 20 fathoms deep, the copepods, according to Peck (1896, pp. 356-368), are about twice as abundant in the more superficial waters as in the middle depths or near the bottom. Those near the top show a remarkable daily rhythm in their movements. During the night they swarm in great numbers on the immediate surface, but from sunrise to sunset they desert the surface almost completely. This change makes itself felt in their availability as a food supply, for Peck (1894, p. 117) has observed that menhaden taken in the daytime contain fewer copepods than those caught at night. Thus the question of what determines the daily migrations of copepods is a problem not only of general interest to the naturalist, but also of practical importance to the fish-culturist.
According to Giesbrecht (1892), Weismann (1877) was the first to suggest a solution for this problem. He believed that light determined the upward and downward migration of pelagic animals and that most animals, being adapted to light of medium intensity, retreated from the surface when the light increased and moved upward when it diminished. An explanation essentially like this was accepted by Fuchs (1882). Chun (1887) opposed this view and maintained that temperature changes were probably the means of inducing daily as well as yearly migrations.
The first to approach the question from an experimental standpoint were Groom and Loeb (1891), who studied the reactions to light of the free-swimming young of barnacles (Balanus perforatus). Their experiments showed that the animals were not constant in their reactions. When placed in a glass vessel in bright daylight they eventually deserted the side of the vessel next the window and swam away from the light, i. e., became negatively phototropic; when, however, they were placed in dim light they sooner or later reversed and swam toward the light, i. e., became positively phototropic. Since the barnacle larvae in the aquarium, where the only changes were in the intensity of illumination, went down by day and came up by night as they did in the open sea, Groom and Loeb believed that this reaction in nature was controlled by light in essentially the same way as it could be controlled in the laboratory. Thus, while they did not deny that temperature changes might have some influence on the movements of the animals, they concluded that light, not heat, was the chief factor in controlling the daily migrations.
In copepods studied by Groom and Loeb (1891, p. 176) no change in reactions to light such as that observed in the barnacle larvae, could be discovered. Subsequently, however, Loeb (1893, p. 96) studied at Woods Hole certain copepods, most of which were probably of the species Temora longicornis; these could be made negatively phototropic by increasing the temperature of the water or by decreasing its density, and positively phototropic by decreasing the temperature or increasing the density. Although Loeb does not state that these factors play any important part in daily migrations, he does call attention (Loeb, 1893, p.105) to gravity as an effective element in this respect. Animals that respond negatively to the force of gravity, i.e., are negatively geotropic, would, so far as this factor alone is concerned, continually move toward the surface.
Thus one of the migratory movements might be accounted for.
From the preceding survey it is evident that the daily migration of pelagic animals is not the result of as simple a combination of circumstances as was at first supposed, and, further, that what may be effective in bringing about migration in
-----------------------------------------------------------
DAILY MIGRATIONS OF COPEPODS.
Page 105
one species may not in another. So far as the very few copepods that have been studied are concerned, the chief factor seems to be light, though reaction to gravity has not been shown to be without influence, and heat and density of the sea water may play subordinate parts.
In attempting to ascertain the factors that determine the migration of copepods, I have carried out a series of experiments on one of the commonest summer species from the region about Woods Hole, Mass., namely, Labidocera aestiva Wheeler (1900, p. 178, fig. 16). This is an inconspicuous copepod of usual proportion, and about 2 millimeters; in length, i.e., a dozen of them placed end to end would measure very nearly an inch.
The males differ from the females in three conspicuous features:
First, the dorsal eye lenses in the male are larger than those in the female; secondly, the right anterior antenna of the male is partly modified to form a grasping organ, while the antennae of the female are unmodified and symmetrical; and, thirdly, the first abdominal segment in the male is simple, and not swollen and covered with hairs, as in the female. This species was found to predominate in the tow taken during the whole of August, 1901. and served in every way as satisfactory material for experimentation.
My work was carried on in the laboratory of the United States Fish Commission at Woods Hole, and i take pleasure in expressing my indebtedness to Dr. H. M. Smith, director of the laboratory, as well as to the station corps, for many courtesies shown me.
MOVEMENTS.
At the outset I put several hundred Labidocerae in a large, cylindrical, glass aquarium nearly filled with sea water and placed on a table near a window. In a short time the Labidocerae had distributed themelves in a verv characteristic way. One set formed a densely packed row at the top of the water on the side of the aquarium next the window; the other was formed of individuals rather uniformly scattered through the whole aquarium. The following enumervtions will suffice to show the composition of these two sets: of 25 individuals taken from the row next the light, 14 were females and 11 males; of 25 caught swimming at large, 23 were males and 2 were females. The almost complete absence of females from the latter set makes it obvious that in experimenting on this species the material should not be considered as homogeneous, but males and females should be dealt with separately.
The movements of the Labidocerae are in part due to the animal's activities and in part to its weight. If a number of females are isolated in a small aquarium and watched, they will be seen sporting about close to the upper surface of the water. From time to time one or other will sink through the water for a distance of an inch or more, and then with a few sudden leaps will regain its former place. The sinking is apparently due entirely to the animal's weight, for it is unaccompanied, so far as can be seen, by the movement of any locomotor organs. The upward leaps are due to vigorous muscular action.
The sinking of the animal through the sea water implies that its specific gravity is greater than that of the water, and such, in fact, is the case. The specific; gravity of the sea water in which this species was ordinarily taken proved on determination to be 1.025, and that of a solution of common salt, which just kept slightly etherized individuals from sinking, was found to he 1.109; the specific gravity of Labidocerae, in reference to sea water, is, therefore, 1.109/1.025 or 1.082. In consequence of this the
-----------------------------------------------------------
BULLETIN OF THE UNITED STATES FISH COMMISSION 1901.
Page 106
animals sink in sea water, and sink at a considerable rate. The time taken by each of 10 etherized individuals to fall through 20 centimeters of sea water varied from 35.2 seconds to 47.2 seconds, and averaged 40.7 seconds. The rate of fall over different parts of the course seems to have been rather uniform, for in a second series of observations the first 10 centimeters were passed over in an average of 21.68 seconds, and the second 10 in 18.58 seconds, giving a general average for the whole 20 centimeters of 40.26 seconds. From this and the preceding observations it is fair to assume that the animals fall through the water at the average rate of about half a centimeter a second. They would thus drop a fathom in about six minutes.
The positions that the animals assume in falling through the water are different in different cases. When they are killed suddenly by adding a small amount of alcohol, formaldehyde, or corrosive sublimate to the water, and are then transferred to ordinary sea water, they descend head downward. When, however, they are placed in water containing a small amount of ether and are thus made motionless without being killed, they fall through ordinary sea water with the head uppermost. This, too, is the position assumed by the living animals when descending. The difference in the mode of descent of the dead and of the etherized animals is due to the disposition of the appendages. Strong killing reagents, such as alcohol, formaldehyde, etc., act so vigorously on the animals that they die with all their muscles contracted. Since the flexors, are stronger than the extensors, the appendages, and particularly the anterior antennae, are folded alongside the body in death and the animal moves head first through the water, probably because of the greater specific gravity of the head.
In the etherized and the normal animals, on the other hand, the anterior antennae are kept spread and the resistance that they meet in their passage through the water retards the falling of the head so that the animal descends with the head uppermost. This is of no small importance to a living Labidocera, for, being thus oriented in its fall, its first efforts at locomotion must be effective in carrying it toward the surface.
Thus, irrespective of other functions that have been ascribed to the anterior antennae, they undoubtedly serve as organs for the orientation of the body. It is not without interest to recall that the function of orientation in the higher crustaceans is also dependent, in part at least, on the anterior antennae, although in these forms this function is connected with a special sense organ, the statocyst, whereas in the Labidocerae the antenme as a whole probably act in a purely mechanical way.
What has been said about the movements of female Labidocerae applies also to the males, except that in their locomotion this sex often does what the other rarely attempts, namely, swims downward. Thus the males gain a much more general distribution than the females.
In both males and females the kinds of locomotion, except for direction, are much the same. Both sexes at times exhibit a uniform continuous movement, as though the animals were creeping rapidly through the water; at other times, and this is the more usual, they pass through the water by relatively enormous leaps, carried out at such high velocities that it is sometimey impossible to follow the animal with the eye. Although intermediate forms of locomotion occur, such as a succession of short leaps, the majority of movements belong to one or other of the two kinds just described.
The leaping movements of the copepods have been usually ascribed to the vigorous action of the anterior antennae. Recently MacBride (1899, p. 505) has called this opinion in question and has maintained, on the grounds of observations on the slower leaping movements, that the leap is effected entirely by the simultaneous action of
-----------------------------------------------------------
DAILY MIGRATIONS OF COPEPODS.
Page 107
the thoracic feet, and that during this operation the antennae are held as rigid as possible at right angles to the chief axis of the body. As MacBride remarks, the extended leaps are carried out with such quickness that the parts taken by different appendages can not be directly observed, and his conclusion, therefore, is based on inference, not on direct observation. But direct observation is not the only means of ascertaining how the leap is accomplished.
It is an easy matter to render copepods quiescent by putting them for a few moments in sea water containing a very little ether. Such animals recover on being placed in ordinary sea water and act in all respects normally. With a sharp-pointed knife it is possible to cut off the anterior antenna and expanded tail ends of etherized Labidocerae, though the other parts are too closely attached to the body to admit of easy removal. Animals from which both anterior antennae and the tail have been removed have been kept a day or more after recovery from ether and their movements observed. They swim with great agility and seem quite as vigorous as normal specimens, but they seldom live beyond 48 hours after the operation. In their locomotion they show only one type of movement, the uniform rapid creeping. This is modified now and then by becoming somewhat uneven, but in no instance have I observed a Labidocera without anterior antennae and tail take even a short leap.
Since in these specimens the thoracic legs are all intact and yet no leaps are made, I feel certain that these appendages are not used in the way that MacBride supposed.
A Labidocera from which the anterior antennae have been removed makes leaps that differ from the normal ones only in being somewhat shorter. If, now, the tail of such an animal is removed, the animal ceases to leap. Thus the tail is unquestionably connected with this method of locomotion. A Labidocera from which the tail has been removed can likewise leap, though in this case, also, the leaps are shorter than the normal ones. When, however, the anterior antennae are also removed the leaping ceases. Thus the anterior antennae, contrary to MacBride's opinion, are, in Labidocera aestiva at least, connected with leaping.
In this species, then, the leap is performed almost exclusively by the combined action of the anterior antennae and the tail; and the thoracic and other appendages, though they may aid it, are not capable in themselves of carrying it out. Since the anterior antennae, and the tail are not in action when the animal is creeping rapidly through the water, this motion must be ascribed to the action of other parts, probably the thoracic appendages.
From the preceding account it might be inferred that the female Labidocerae retain their positions at the surface of the water and the males keep from accumulating on the bottom by incessant muscular activity, whereby they lift themselves against gravity; but, though this is in large part true, it is not entirely so, for it is a most usual occurrence to find both males and females attaching themselves to fixed objects, such as the sides of a glass aquarium. This they can do even when the glass is so tilted that they are well on the under side of it.
If such a suspended animal be approached by a sucking tube the current of water that can thus be produced may be made to swing the animal back and forth on its support, and it can then be seen that the only parts in contact with the glass are the anterior antennae. Not only is this so, but a careful adjustment of the strength of the current will often partially loosen the animal, so that for a while it hangs in the water with an attachment to the glass by only the tip of one anterior antenna. It is really remarkable with what success an animal thus almost completely loosened from the glass will still retain its hold. That the other appendages are not concerned in thus anchoring the animal is
-----------------------------------------------------------
BULLETIN OF THE UNITED STATES FISH COMMISSION 1901.
Page 108
made clear in two ways: First, when the animal hangs from the underside of a piece of glass the main axis of the body is nearly vertical, and thus the other appendages are not in a position to touch the glass; and secondly, when animals anchor themselves to vertical surfaces of glass they do so quite as frequently by the dorsal surface of the body as by the ventral, and, since their appendages other than the anterior antennae are on the ventral side, it follows that when they rest with the dorsal surface to the glass the only appendages that can touch the glass are the anterior antennae.
How the antennae adhere to the glass is not certainly known, but it is quite probable that the attachment is accomplished by means of the numerous hairs that cover these organs and that are especially abundant at the tip. (Wheeler, 1900, p. 179, fig. 16c.)
Not only can Labidocerae anchor themselves hy their antennae to fixed objects in the water, but they can likewise hold to the surface of the water. A jar of water containing many Labidocerae can be so placed that the animals may be seen hanging from the surface of the water exactly as they would hang from the under surface of a piece of glass. To the surface of the water as to the glass the animals are attached by their anterior antennae. Slight waves are sufficient to dislodge them, but in quiet water great numbers hang in this way for considerable periods.
The mechanism of attachment in this as in the former case has not been definitely ascertained, though it is very likely, as Scourfield (1900, p. 307) has suggested, that the hairs of the anterior antennae pierce the surface film of the water and produce capillary depressions by which the weight of the body may be supported.
Thus, Labidocerae, whose weight ordinarily causes them to sink in sea water, overcome this tendency by gliding movements involving the action of their more centrally placed appendages, by leaps accomplished through the combined action of tail and anterior antennae, and by attaching themselves through the anterior antennae to fixed objects in the water or to the surface film. In these operations the anterior antennae perform at least three functions: They orient the animal mechanically as it falls through the water, they are organs of positive locomotion in connection with the leaping habit, and, finally, they are organs of attachment while the animal rests.
REACTIONS TO GRAVITY (GEOTROPISM).
As already pointed out, most female and many male Labidocerae frequent the top of the water in any large aquarium much as they do the top of the sea, and, as we have seen, they are continually falling from this situation because of the influence of gravity and as continually exerting themselves to regain it. Do the Labidocerae move toward the surface because it is nearer the source of light, i.e., are they positively phototropic, or do they move in that direction because it is away from the earth's center of gravity, i. e., are they negatively geotropic?
To test this question a large glass tube about 8 centimeters in diameter and some 60 long, with one end open and the other closed, was set up vertically and nearly filled with sea water. On the side of the tube, and beginning at the bottom, five intervals of 10 centimeters each were laid off and marked, and the water in the tube was made to stand at the 50-centimeter mark. The apparatus was so arranged that the tube could be inclosed in a light-proof jacket without disturbing the contents by jarring, etc.
The first experiment consisted in putting five female Labidocerae in the tube and recording their distribution under the influence of diffuse daylight. The sections
-----------------------------------------------------------
DAILY MIGRATIONS OF COPEPODS.
Page 109
in the tube were numbered from the bottom up, I to V, and the records were taken at the ends of five-minute intervals till ten records were obtained. The results are indicated in Table 1, where it appears that in eight records out of ten all five animals were in the uppermost section, and that in two records one animal in each case had descended to the fourth section:
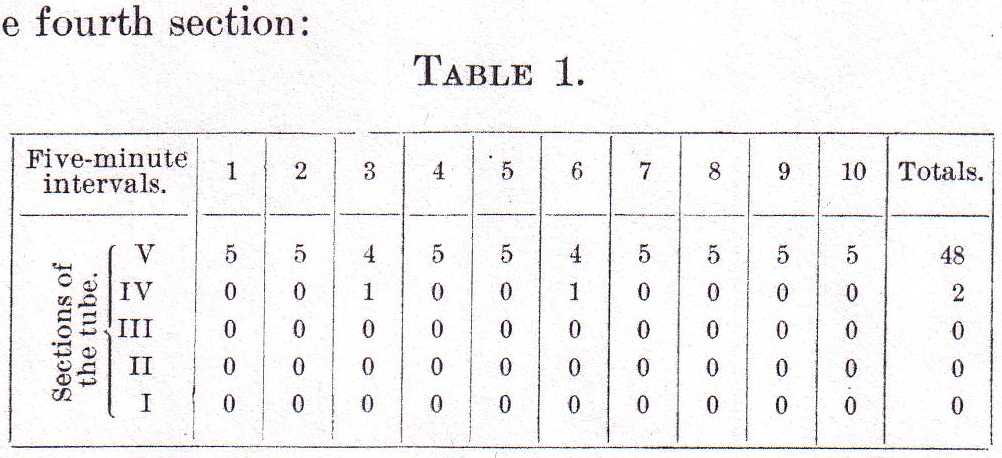
To ascertain how much influence light had in bringing about this condition, the tube with its contents was now placed in perfect darkness and exposed for a moment to light every five minutes to record the distribution of the Labidocerae. Table 2 shows the results of this experiment.
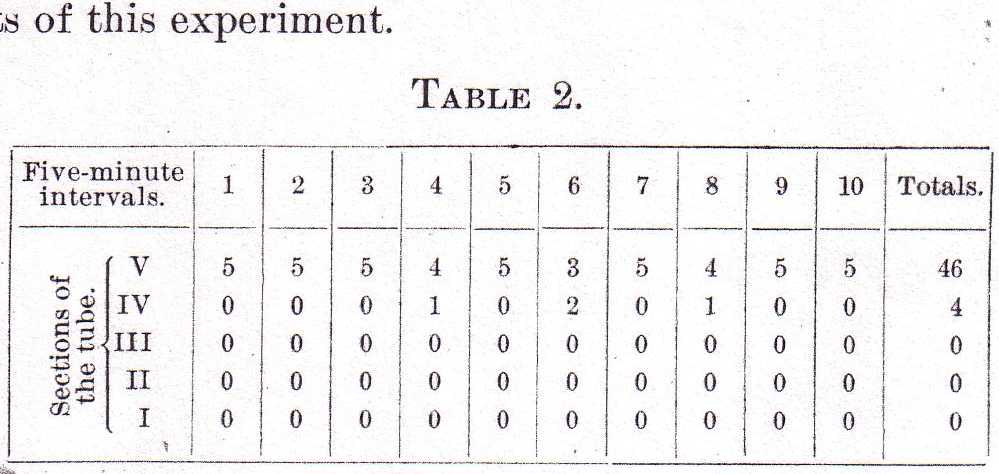
Here in seven records out of ten all five animals were in the uppermost section; in the remaining three records four animals were in the fourth section. Thus the record in Table 2, though not quite so nearly uniform as in Table 1, differs so little from the latter that the conclusion that female Labidocerae stay at the top of the water because of some other stimulus than light is entirely justifiable.
Although, next to light, gravity is the most probable factor in determining the distribution of female Labidocera, it might be assumed from anything that has thus far been shown that these animals seek the surface because of the greater abundance of oxygen there. To test this assumption a large glass tube, such as that described on page 108, was filled with sea water and inverted in an aquarium in such a way that no air entered it. Five female Labidocera were introduced at the lower open end; they swam rapidly to the upper end, where they were, of course, at the top of the water, but without being, in contact with the air. Here they remained till their deaths stopped the experiment. The first one died 68 hours after introduction; the last one 85 hours.
As the animals remained persistently at the upper closed end of the tube, it is evident that the atmosphere is not a factor in determining this feature of their distribution. If, then, neither light nor atmosphere is essential in keeping female Labidocerae at the surface of the water, the only other probable factor is gravity, and, as these animals persistently work against gravity in keeping near the surface, their geotropism is negative.
-----------------------------------------------------------
BULLETIN OF THE UNITED STATES FISH COMMISSION 1901.
Page 110
With the male Labidocerae the case is quite different. First of all, they can be divided into two classes--those that are found at the surface with the females and those that are scattered through the water. Individuals from both these classes were subjected to experiments such as have been already described for the females. Table 3 shows the distribution at five-minute intervals of five males taken from the class of scattered individuals and open to the action of both light and gravity, as in the first experiment with the females (Table 1).
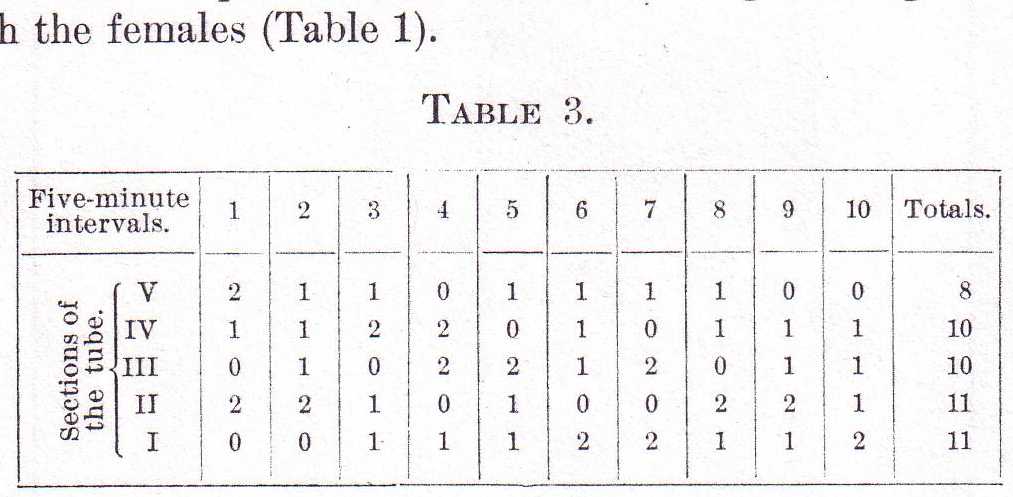
The distribution as shown in this table indicates almost complete indifference, as might have been expected from the previous distribution of these animals in the water. Table 4 shows the results from the same five males when the tube was placed in the dark, as in the second experiment with the females (Table 2).

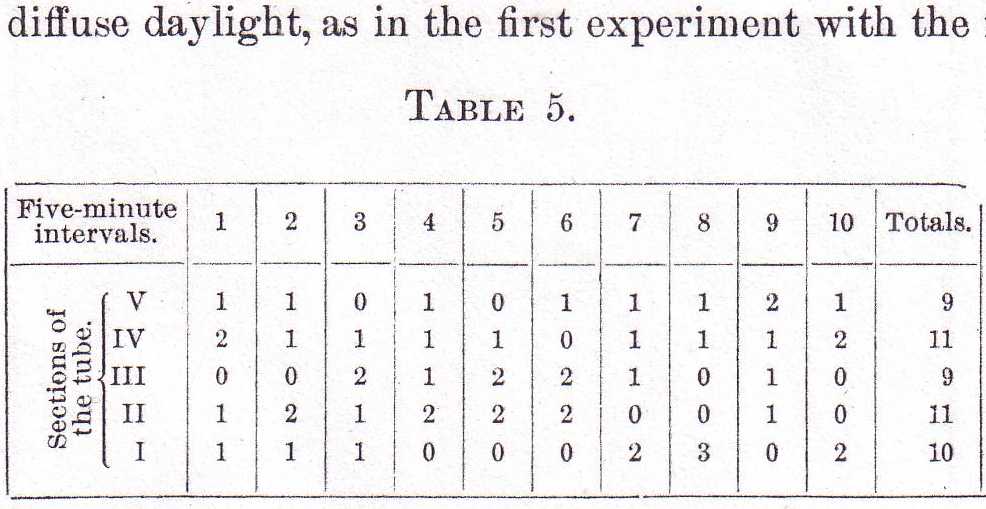
The results shown in this table are so nearly like those in Table 3 that it is safe to assume that light plays no important part in the distribution of this class of males. The second class of males, those found at the surface with the females, might naturally be expected to show the same reactions to light, gravity, etc., that the females do. This, however, is far from the case. Table 5 gives the distribution of five males taken from among the females at the top of the water, and placed by themselves in a glass exposed to diffuse daylight, as in the first experiment with the females.
-----------------------------------------------------------
DAILY MIGRATIONS OF COPEPODS.
Page 111
It is evident from this table that the separation of the males from the females totally changes the plan of distribution of the males. They become scattered more or less uniformly, as in the case of the first class of males. They likewise are undisturbed in their new distribution by the absence of light, as is shown in Table 6, in which the distribution of the same five males used for Table 5 is recorded, but with the difference that the tube was kept in total darkness except for the brief periods of observation.
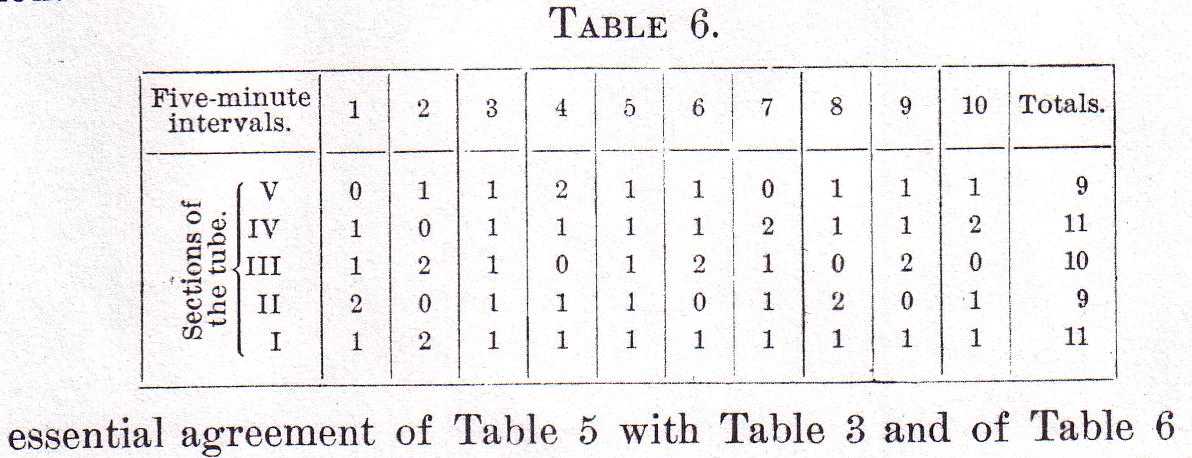
The essential agreement of Table 5 with Table 3 and of Table 6 with Table 1 shows that all males react or fail to react to light, gravity, etc., in much the same way, and that it is the responses to the females rather than to other factors that divide the males into two classes. So far as their reactions to light, gravity, etc., are concerned, all males form in reality only a single class.
It must be evident from the preceding experiments that light is a very subordinate factor in determining the distribution of the males, if, in truth, it is to be reckoned with at all, and it might further be supposed, since the males when left to themselves do not collect either at the top or at the bottom of the aquarium, that they therefore are also uninfluenced by gravity, i. e., that they are not geotropic.
This conclusion, however, would be erroneous, in my opinion, for the bodies of the males are quite as heavy as those of the females, and are continually falling toward the bottom. If the males did not react against this, i.e., were slightly negatively geotropic, they would eventually all reach the bottom, a form of distribution which we know does not occur. I therefore believe that the males also are negatively geotropic, but only sufficiently so to keep them from collecting at the bottom and not enough to cause them to collect at the top, as the females do.
Two conclusions may now be regarded as well established: first, that the female Labidocerae are strongly negatively geotropic, and, secondly, that the males are slightly so. The first of these makes clear why under natural conditions the females come to the surface of the sea and stay there; neither helps us to understand why the males congregate with the females or why the latter at stated intervals desert the surface of the sea.
Loeb (1893) long ago pointed out that by appropriate stimulation the sense of an animal's response might be inverted; thus an organism positively phototropic under ordinary condition might by a slight change in its environment become negatively phototropic. It is therefore necessary, in attempting a further insight into the daily migrations of the copepods, to ascertain whether there are not means at hand for converting the negative geotropism of the females into a positive geotropism, thus establishing a sequence of events that might offer an explanation of daily migration. Heretofore some of the most successful means in accomplishing such inversions have been mechanical stimulation, as agitation of the water in which the animals are, and changes in the density and in the temperature of the sea water.
-------------------------------------------------------------------------
BULLETIN OF THE UNITED STATES FISH COMMISSION.
Page 112
Both the male and the female Labidocerae, after the rough handling incident on being squirted several times from a pipette into a vessel of sea water, still showed unchanged their characteristic geotropic responses; and even after they had been violently shaken in a test tube partly filled with sea water no alteration in their geotropism was observed. Mechanical stimulation, therefore, seemed insufficient to change their reactions to gravity. This accords with what was observed on the surface of the sea, for at night Labidocerae were often as abundantly obtained when the sea was rough as when it was smooth.
Changes in density were next tried. Sea water of a specific gravity 1.025 was filtered, concentrated by boiling, cooled, and aerated, and then diluted with distilled water till the requisite densities were obtained. Females introduced into waters of specific gravities 1.050 and 1.035 retained their negative geotropism and showed no differences from normal animals except that they moved more frequently by leaps. Males when placed in similar concentrations frequented the upper portion of the vessel more generally, i.e., had their negative geotropism somewhat increased, and leaped more frequently. Concentrated sea water then induced leaping movements in both sexes, increased slightly the negative geotropism of the males, without, however, making any noticeable change in the geotropism of the females.
In sea water of less density than the normal, the reactions of the males and the females, unless otherwise stated, were similar, and the following records apply to both sexes. In sea water diluted from 1.025 to 1.020 the animals darted about when first introduced and then gradually became distributed as was usual, the females near the top and the males scattered. In water of the specific gravity 1.015 the animals introduced darted about vigorously, after which some went to the bottom, 50 centimeters distant, and some remained above. In water of a specific gravity 1.010 all animals exhibited darting movements and went to the bottom in irregular spirals. They were left in the mixture, and all died in the interval between a half hour and an hour after introduction. In water of a specific gravity 1.005 all fell rapidly, each making one or two leaps and reaching the bottom apparently dead.
In pure water the animals made one or two leaps, and were dead before they reached the bottom. The effect of diluting the sea water did not seem in any case to change geotropic responses. Slight dilution stimulated the Labidocerae to leaping, and greater dilutions, after stimulating them to a few leaps, rendered their movements incoordinate, so that they fell to the bottom without showing responses which at any time could have been interpreted as a change in the sense of their geotropism.
When ordinary sea water was covered by an inch or so of sea water diluted to a specific gravity 1.015 and Labidocerae were liberated at the surface, they almost always sank with a few violent leaps till they entered the ordinary sea water, whereupon they became normally distributed, in that most of the males dispersed and the females congregated near the upper surface, not of the water as a whole, but of the normal sea water.
This experiment indicates that under natural conditions even excessive rain is not likely to change essentially the distribution of Labidocerae, for they would at most be driven only a few inches from the surface. Dropping fresh water into a tube of sea water containing female Labidocerae, so that in the course of about an hour the level of the tube rose a centimeter, could not be said to have driven the animals from the surface, though they occupied a somewhat deeper position at the
-----------------------------------------------------------------------
DAILY MIGRATIONS OF COPEPODS.
Page 113
end of the experiment than at the beginning. The exposure of a similar tube to an actual shower of rain on a very dull day was followed by no observable change in the distribution of the animals. Rain, therefore, with the slight accompanying change in the density of the surface waters, plays no part worthy of consideration in the distribution of Labidocerae.
Temperature changes were next tried. In testing the effects of these changes, animals were used that had been kept several days in a large glass aquarium, whose temperature was uniformly 23° C. Five male Labidocerae were transferred from the general aquarium to the large glass tube described on page 108, now filled with sea water at 23° C. to the 50-centimeter mark. The animals were soon scattered through the water, as characteristic of males. They were now carefully taken out, and the water changed for some at 15° C. On reintroducing the males, they again gradually scattered through the water. The same was true for water at 10° C., as well as for that at 30° and 35°. These temperature changes, therefore, did not alter the distribution of male Labidocerae.
Five females were then subjected to a series of corresponding experiments. At 23° C. the females remained near the top, and the same was true at 15° and 10° C. At 30° C. and at 35° C. all the animals swam rapidly downward to the bottom. To ascertain whether this reaction was in any way connected with a possible change of the animals in reference to light, the experiments were repeated in the dark. Five females were introduced into the tube containing water at 30° C. and the tube placed in a light-proof chamber. After 10 minutes the tube was inspected, and all five animals were found at its bottom.
There thus seems to be no question but that an increase of temperature over the normal converts the geotropism of female Labidocerae from negative to positive.
Something of the nature of this change may be inferred from the following experiments. Five negatively geotropic females were introduced into a large vessel filled with sea water at 30° C. They immediately became positively geotropic and swam to the bottom. After having remained there some minutes they were transferred by a pipette to the bottom of a second vessel filled with sea water at 24° C. They immediately became negatively geotropic and swam to the top. The change from one condition to the other, as these experiments showed, takes place almost instantly, and the condition lasts only as long as the appropriate stimulus is present.
This may be illustrated by another experiment. The lower half of a large glass tube was filled with sea water at 24° C., the upper half with sea water at 30° C. The warmer water was poured into the tube so as to mingle as little as possible with the cooler water. A female Labidocera was now introduced at the top to ascertain whether she would swim to a point deeper than the separation between the waters of different temperatures. She swam rapidly downward, but stopped almost exactly at the plane of separation for the two temperatures.
A second tube was prepared containing sea water at 21° C. in the lowest third, at 23° C. in the middle third, and at 35°C. in the uppermost third. In this tube a thermometer was hung by a thread and a female Labidocera introduced at the top. She swam down almost at once to a point near the middle, which, the thermometer having been adjusted, was found to have a temperature of 23° C.; she then rose somewhat, and five minutes after introduction was in water of 26° C. Ten minutes later she had risen somewhat farther, but was still in water registering 26° C. After half an hour, when the water in the top of the
-------------------------------------------------------------------------
BULLETIN OF THE UNITED STATES FISH COMMISSION 1901.
Page 114
tube was at 26.5° C., she was still some inches from that level and in water at 26° C. It would therefore seem that 26° C. is the temperature limit above which this female Labidocera became positively geotropic.
That there should be no question about the difference between the males and the females in their capacity to be transformed geotropically, the following experiment was tried. Ten animals taken at random from the top of the general aquarium were suddenly introduced with as little water as possible into a tube filled with sea water at 30° C. They quickly separated into two sets, one going to the bottom, the other remaining near the top. The six that remained near the top were picked out and all proved males; the four that swam to the bottom were then recovered and found to he females.
These observations show that the temperature of the water has no obvious effect on the slight geotropism of the males. The geotropism of the female is negative in cold water and positive in warm, and for normal animals the critical temperature seems to he about 26° C.
It thus appears that of the three means suggested whereby the sense of the geotropism of Labidocerae might be changed, only one, namely, temperature, has proved really effective. The critical point in the temperature change is, however, so high, compared with any temperature range the animal is likely to meet with naturally, that the assumption that the daily migrations of the females depend on geotropism changed from negative to positive and back again by temperature differences is wholly unwarranted. So far as one can see, the only part played by geotropism in the daily migrations affects the females; they rise to the surface and stay there because of their strong negative geotropism.
REACTIONS TO LIGHT (PHOTOTROPISM).
Light, like gravity, influences bodies of water, as a rule, from one side only, and, since it increases and decreases during the day, it has naturally been regarded as a potent factor in determining the daily migrations of pelagic animals. That it plays a part in the distribution of copepods can be seen from the fact that in an aquarium standing near a window many Labidocerae congregate near the surface of the water on the side next the window; i. e., they are positively phototropic.
If a considerable number of male Labidocerae are placed in a large glass jar filled with sea water and illuminated from one side, they will soon be found scattered through the water. A close inspection, however, will show that rather more are in the half of the jar away from the light than in the half toward it.
To determine with greater accuracy what this difference amounted to, a nearly cubical glass jar filled with sea water was so arranged that a temporary glass partition could be slipped into it vertically, thus dividing its contents into a front and a back half. One meter in front of the jar was placed an incandescent electric lamp of about 14 candle power.
The light from this fell perpendicularly on the front face of the jar and the partition was so placed that of the two chambers formed by it one was toward the lamp, the other away from the lamp. The partition having been withdrawn, ten male Labidocerae were liberated in the middle of the jar and after ten minutes exposure to the light the partition was inserted and the numbers in the chamber toward the light and in that away from it were counted. The experiment was then repeated, the Labidocerae being each time liberated at the center of the jar and allowed ten minutes in which to become distributed. Table 7 gives the result of ten such trials.
-------------------------------------------------------------------------
DAILY MIGRATIONS OF COPEPODS.
Page 115
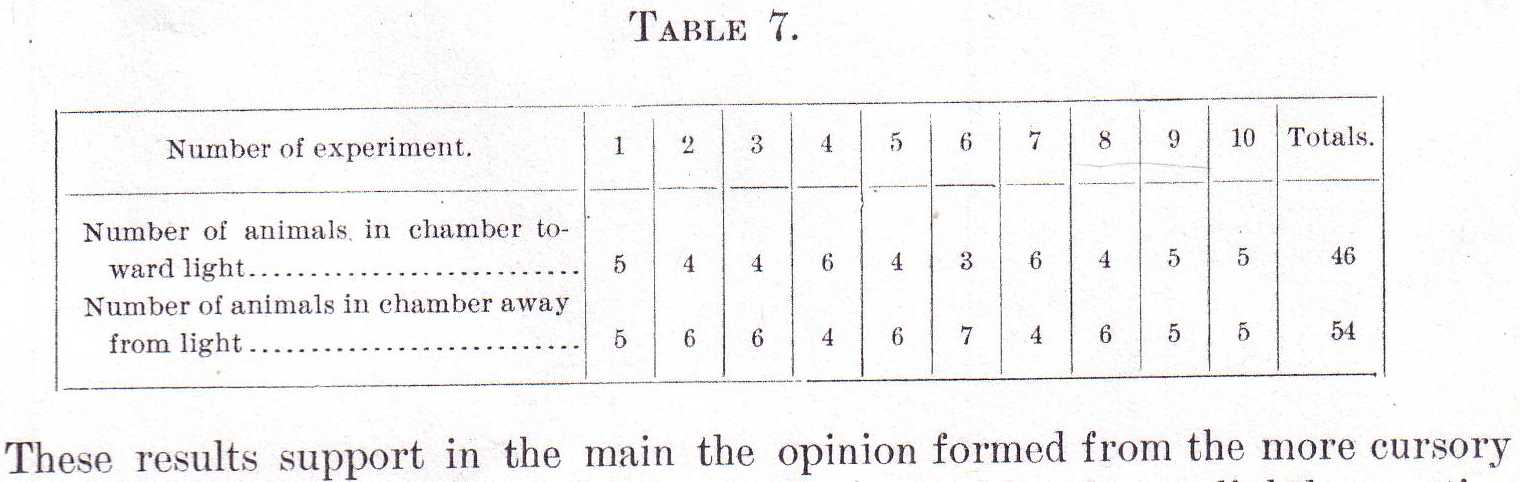
These results support in the main the opinion formed from the more cursory inspection of the jar. As a whole the assemblage of males shows slightly negative phototropism. These experiments do not, however, preclude the possibility that some individuals are slightly positive and some indifferent; they merely show that more individuals are slightly negative than otherwise.
When a single male is put in a large open glass jar, the animal can usually be driven about from one side to the other by holding an electric light (14 candle power) close to the jar. The reaction is never very quickly performed and sometimes fails entirely, but usually after a few minutes the animal swims from the illuminated side to the opposite one and stays there persistently. Although it is generally not difficult to drive a male Labidocera by light back and forth horizontally through a jar, I found it almost impossible to drive them up or down through a thickness of water equal to that through which they would move horizontally. Sometimes this seemed to succeed, but generally not, and I was finally forced to conclude that, slight as the geotropism of the males was, it was more effective than their phototropism.
Female Labidocerae collect on the light side of a jar with great precision. Since they are also negatively geotropic they keep close to the top of the water. When ten females were exposed to light in the square glass jar with the partition as described for the males (p. 114), all were found in all ten trials in the chamber next the light. Their positive phototropism is thus clearly indicated.
Further evidence of this peculiarity was observed in several ways. The large upright glass tube marked off at 10-centimeter intervals (p.108) was filled with seawater to the 50-centimeter level and five females were placed in it. The whole was exposed to diffuse daylight in a room, and the Labidocerae soon congregated at the top of the water on the side of the glass nearest the window. An opaque cover was now slipped over the top of the tube and down to the 40-centimeter mark. In a few moments all five animals had dropped to this level, and remained here as long as the cover was kept in this position.
This result was essentially the same as that observed by Loeb (1893, p.160) for Temora longicornis, except that in Loeb's experiments the copepods were checked in their upward movements by the opaque cover instead of being driven downward by it. In either case it shows that the animal's positive response to light is stronger than its negative response to gravity. By lowering such an opaque hood over the tube the Labidocerae were driven to the bottom of the tube, where they remained as though imprisoned. They retained this position as long as a small amount of light was allowed to enter at the bottom, but the moment this was cut off they presumably began to rise, for on removing the hood ten minutes later they were all found at the top of the water. Thus their negative geotropism is at once made effective by complete darkness.
-------------------------------------------------------------------------
BULLETIN OF THE UNITED STATES FISH COMMISSION 1901.
Page 116
The rates at which some of these reactions were carried out are not without interest. In one set of experiments the females were made to descend 15 centimeters from the surface by means of the opaque hood. From the time the hood was put on, which presumably marked the beginning of the descent, till the arrival of the animals below, was exactly 30 seconds. This indicated a descent at the rate of half a centimeter per second, which has already been shown to be the rate of passive falling.
So far as one could judge from the first sight of the animals as they emerged below the level of the hood, they did descend by falling and not by any form of positive locomotion. It would, therefore, seem that diffuse daylight from below acts on female Labidocerae rather as an inhibiter of their negative geotropic reactions than as a positive phototropic influence. On the removal of the hood the females returned to their original position, but required 1 and one half minutes to perform this migration. They thus descended three times as rapidly as they ascended--i.e., if they can descend a fathom in about 6 minutes they could probably not make that ascent in less than 18 minutes.
In another experiment five female Labidocerae were put in a glass tube, of large caliber and about 50 centimeters long, and hung vertically in a dark chamber. When an incandescent electric lamp (14-candle power) was brought near the side of the tube, the five animals crowded close to it.
When the lamp was held at the top they all ascended to the top, and they could easily be induced to descend through 50 centimeters of water by holding the lamp under the bottom of the tube. Their descent was accomplished chiefly by passive falling, with the anterior end uppermost, though now and then they swam downward, an unusual action for a female. In this experiment, as in former ones, the responses of the females to gravity were always subordinate to those to light--i.e. geotropism was less pronounced than phototropism.
A curious relation between these two sets of reactions was seen in the following experiment. The large tube, graduated to 10-centimeter intervals, was filled with sea water to the 50-centimeter mark; into it were put five females and the apparatus was covered with an opaque hood from the top to within 5 centimeters of the bottom.
In this lowest section of 5 centimeters all the females congregated. The hood was now taken off in sections, beginning at the top. First, the top with 5 centimeters of the side covering was removed, leaving the tube covered for a distance of 40 centimeters, beginning at 5 centimeters from the bottom. On thus illuminating the interior of the tube from above, it might have been expected that the five females would have risen through the dark section to the now light top; they, however, remained at the bottom. The uppermost 10 centimeters of the side covering were next removed, leaving 30 centimeters still covered, but without effect on the animals.
The next lower 10 centimeters were now taken off, leaving 20 centimeters covered, whereupon four of the Labidocerae immediately swam upward through the dark section to the light at the top. On removing the next 10 centimeters of the side covering the fifth animal ascended. Thus it appears that a dark region between a light top and light bottom may prevent female Labidocerae from ascending, an operation, however, that they may perform before all the dark section has disappeared.
The experiments on light thus far described make it clear that the phototropism of the male Labidocerae is at most slightly negative, that of the females strongly positive. So far as these facts are concerned they seem to have little bearing on daily migrations, for the slight negative phototropism of the males is not sufficient to overcome
-------------------------------------------------------------------------
DAILY MIGRATIONS OF COPEPODS.
Page 117
their negative geotropism and the positive phototropism of the females might be expected to bring them to the surface of the sea in the day instead of in the night, as is their habit.
The positive phototropism of the females is, however, so strong that the question arises here, as it did in connection with geotropism, Are there not perhaps natural means of inverting this? Certainly the conditions in the laboratory did not reproduce those in the outer sea water, for in a large jar kept on the laboratory table the female Lahidocerae kept persistently near the top of the water day and night, although in another large jar floated in the outer basin of the Fish Commission dock, and thus freely exposed to the elements, the females made regular migrations, being close to the surface in the night and at the bottom in the day.
Experiments were now undertaken to ascertain whether the sense of the phototropism of the females or possibly even of the males could be changed. Loeb (1893, p. 96) found that for the copepod Temora longicornis an increase of temperature changed positively phototropic individuals into negatively phototropic ones and intensified the negativity of negative specimens, and that a decrease of temperature changed negative into positive individuals and increased the positivity of positive ones. Holmes (1901) found that in certain amphipods an increase of temperature certainly hastened and perhaps induced the positive condition. Yerkes (1900, p. 417) was unable to change the sense of the light reactions of Daphnia and of Cypris by temperature differences. All efforts to change the phototropism of Labidocerae by changing the temperature of the sea water were without avail. Both males and females taken from water at 23° C. and placed in waters at 10°, 15°, 30° and 35° C. remained, so far as their phototropism was concerned, unchanged.
Density differences were next tried. Loeb (1893, p. 97) had shown that in Temura dilution of the sea water made positive individuals negative, concentration the reverse, but in Labidocera, animals taken from sea water of a specific gravity 1.025 and placed in waters of 1.050, 1.035, 1.020, 1.015, 1.010, 1.005, and 1.000 specific gravity showed no phototropic changes.
Loeb (1893, p. 96) noticed that when Temora was first caught, it was often positively phototropic, though under ordinary circumstances it was negatively so. Shaking made the animals temporarily positive, and probably explains the peculiarity just mentioned. Towle (1900), by a most painstaking series of experiments, showed that Cypridopsis was usually negatively phototropic, but that contact with a pipette and other slight mechanical stimulations were sufficient to make it temporarily positive.
Conditions parallel with those in Temora and in Cypridopsis were often found in Labidocera. Females are ordinarily positively phototropic, but after having been several times vigorously ejected from a pipette into sea water, the majority of them become temporarily negative.
The following laboratory record will show clearly the nature of this change.
Aug. 20 (1901), 7.35 p.m. Five positively phototropic females were ejected, each one three times, from a pipette into sea water. Two remained positive and three became negative, swimming to the side of the glass vessel away from the light.
7.45 p.m. One of the three negative individuals became positive and swam with some irregularity to the two positive individuals on the light side of the vessel.
7.58 p.m. The second negative individual became positive.
9.20 p.m. The third and last negative one became positive.
-------------------------------------------------------------------------
BULLETIN OF THE UNITED STATES FISH COMMISSION.
Page 118
Since females can be rendered negatively phototropic for a period of almost two hours by handling them with a pipette, it might he inferred that this is due to simple mechanical stimulation. That such is not exactly the truth is seen in the fact that female Labidocerae agitated in the water do not become negative even when this process is several times vigorously repeated, although these same animals easily change on being handled with a pipette. Apparently, then, it is a very special form of mechanical stimulus that is necessary to call forth this reversal.
Pipette experiments with live males, which in the beginning were almost indifferent or only slightly negative to light, resulted in nnaking all distinctly negative, a condition which lasted at most an hour and a quarter.
Although certain forms of tactual stimulation are a means of making both males and females move away from a source of light, even, as I found by experiments, downward through the water, they can not for a moment be regarded as the efficient cause in the downward movements of the Labidocerae each morning, for the animals are quite as much open to agitation at other times of day as in the early morning.
Notwithstanding that this change has probably no connection with the daily migrations, it may have a very important connection with the pairing habits of the animals. As is well known, the right antenna of the male is modified to form a grasping organ, and by means of this the male seizes the female in pairing and holds her till he has deposited a spermatophore on the posterior part of her body.
I have observed pairing among Labidocerae a number of times, and it has always taken place between individuals at the top of the water. After the male has seized the female, the pair plunge about in a most violent manner, and it has occurred to me that the mechanical stimulation thus produced might he sufficient to render both negatively phototropic, so that after release they would move downward to the water where fewer individuals were. This operation, if it really takes place, would have the effect at least of removing from the pairing individuals any two that had recently been in union.
After this idea occurred to me, I observed only one pair in copulalation. These were removed from the general aquarium to determine, whether after separation their light reactions had changed. After separation, however, hoth male and female exhibited their usual phototropic responses, no change having taken place, but on examining the female no spermatophore was found on her, and possibly the operation was incomplete in respect to light as it was in sexual matters.
Attempts were next made to ascertain whether the continuance of light action or a difference in its intensity could induce, changes in the Labidocerae. Groom and Loeb (1891) observed that the free-swimming larvae of a barnacle were positively phototropic in the morning, but that before noon and without other changes than continued light action they became negative. These in the course of the night all became again positive. Thus the continued action of bright light gradually made the larvae negative, of dim light positive.
To ascertain the effect of this form of continuous light action on Labidocerae, I put five males in one large jar of sea water and an equal number of females in another. These were exposed to diffuse daylight in the laboratory, as in Groom and Loeb's experiment, the jars having been set up August 12. On August 20 one male died,the other four succumbed between August 24 and 26. One female died August 24,
-------------------------------------------------------------------
DAILY MIGRATIONS OF COPEPODS.
Page 119
and the experiment was discontinued August 26. During all this period the males and females retained their characteristic distribution, and nothing whatever suggestive of daily migrations was observed. Thus the changes noticed by Groom and Loeb in barnacle larvae failed entirely to appear in Labidocerae.
Lights of different intensities were next tried. When an electric incandescent lamp of about 14-candle power was held about 10 centimeters from a glass vessel containing female Labidocerae, the animals came to the side next the light; they likewise went to the top or to the bottom of the vessel, depending upon whether the light was held above or below it. These same reaction, obtained when a concentrated solution of alum was used as a means of excluding heat rays. The reactions are therefore due to light.
When, in place of a 14-candle lamp, one of about 100-candle power was used, the results were very different. The Labidocerae, on first being approached by the intense light, made one or two leaps and then swam directly away from the light. If the lamp was placed above them they moved to the bottom of the aquarium (about 30 centimeters); if, now, the light was quickly brought under the vessel, the animals rose. This latter response was doubtless coupled with their negative geotropism.
Thus it is obvious that female Labidocerae are positively phototropic to light of low intensity, and negatively so to light of high intensity. (See footnote *)
The phototropism of the males was not noticeably different in the intense light from what it was in the dim light. Under both conditions they were, its a rule, slightly negative.
These observations make clear why the female Labidocerae are not at the surface of the sea in the daytime. They are negatively phototropic to its bright light, and therefore pass downward to the deeper water, where, from absorption by the upper layers, the light has decreased in intensity.
To demonstrate the validity of this conclusion, a jar of female Labidocerae was covered with a glass dish containing 4 inches of a saturated alum solution to absorb heat rays and then exposed to direct sunlight about noon. Immediately on being put in the sunlight the animals retreated from the source of light and went to the bottom of the jar. In this respect they were like Simocephalus sima as observed by Radl (1901, p. 83). On excluding sunlight from the jar the Labidocerae again rose, and they could be thus driven up and down at will.
The migrations thus produced undoubtedly imitated in miniature the natural daily migrations of female Labidocerae in the sea. These animals migrate into deeper waters on the coming of daylight because of their negative phototropism to strong light. They return to the surface on the approach of night because of their positive phototropism for weak light and their negative geotropism, a combination which Loeb (1890, p. 42) has already shown in the larvae of the brown-tail moth.
In this way the migrations of the females are accomplished, and the only reason why these animals do not carry out similar daily movements in the laboratory is because of the absence of one factor, intense light.
(Footnote:*) Conditions of this kind, so far as reactions to light intensities are concerned, were apparently first noticed by Famintzin (1867) in the protozoans Chlamidomonas and Euglena, and have since been observed in the following animals: Hydra (Wilson, 1891), Polygordius larvae (Loeb, 1893), Daphnia, Lubbock, 1884), and Limax (Frandsen, 1901).
-------------------------------------------------------------------
BULLETIN OF THE UNITED STATES FISH COMMISSION 1901.
Page 120
Although this explanation makes clear the movements of the females, it in no wise applies to the males, for, as we have seen, they are almost indifferent to the effects of light and gravity. The occasion of their migrations must be sought for in other factors.
REACTIONS TO SUBSTANCES IN THE WATER (CHEMOTROPISM).
The Labidocerae that collect at the surface of the water on the light side of an aquarium have already been shown to be some males and some females. A hundred or more of these were placed in an aquarium filled with sea water and in a short time the majority of the newcomers were found at the top of the water near the light; others, however, were scattered throughout the whole body of water. These scattered individuals were then picked out, and, as might have been expected, were found to be almost exclusively males. An hour or so after this the aquarium was again examined and was apparently as full of scattered males as before.
These, like their predecessors, were removed. After a time, however, they seemed to reappear, although in diminishing numbers. The process of picking out was continued until few or no scattered individuals could be found. An examination of the assemblage at the top now showed it to be made up almost exclusively of females, the males having escaped for the most part to the lower water, where they were picked out.
Under ordinary conditions, then, males are continually escaping from the assemblage of males and females at the surface and swimming off to become scattered individuals in deeper water.
The males not only are continually leaving such an assemblage; they are also continually entering it. At the bottom of a glass aquarium containing only female Labidocerae ten males were liberated. They swam about as scattered individuals for some time, but after half an hour only six could be seen; four, as a close inspection showed, had joined the assemblage of females at the top. The females must, therefore, be admitted to have some power of attracting the males.
It occurred to me that the possible phosphorescence of the female might, at least at night, be a means of attracting the males. Among the abundant phosphorescent organisms in the summer waters at Woods Hole small luminous points had often been shown me as phosphorescent copepods. By taking at random test tubefuls of sea water at night a single such phosphorescent point could be isolated. Such samples of sea water contained sometimes one, sometimes more copepods, but they were so frequently without copepods at all that I was forced to decide that the luminous points were something other than these animals. Moreover, when a single male or female Labidocera was put in a test tube containing sea water which on previous agitation showed no phosphorescence, and the whole vigorously shaken, not the least phosphorescence was ever observed. Labidocerae aestiva is, in my, opinion, not phosphorescent, and therefore this factor can have no part in causing the males to be attracted by the females.
Since phosphorescence seems to play no part, and since the higher sense organs of the Labidocerae, the eyes, etc., are so very rudimentary, some form of scent would seem the most natural way by which the males might be led to the females. This would probably depend upon some substance given out by the females to which the males would react. That there probably is such a substance is seen from the following
----------------------------------------------------------------
DAILY MIGRATIONS OF COPEPODS.
Page 121
experiments. A piece of glass tube about 2 centimeters long was filled with sea water, its ends were plugged with wet cotton, and it was wrapped in a little more than one fold of white filter paper. A thread was tied around the tube, thus holding the filter paper in place, and the apparatus prepared in this way was lowered into the surface water of a small aquarium containing five male Labidocerae. These swam freely to and fro, and now and then collided with the tube.
They were watched accurately for thirty minutes, and the number and character of the collisions noted. In all, ten collisions occurred, after each of which the animal usually deserted the tube at once. Those animals that just missed the tube in passing went on by it without special reaction. The tube was now withdrawn from the aquarium and five females were introduced into it. Because of the filter paper and the plugs of cotton these were not visible from the outside. The tube was again placed in the aquarium, and after five minutes the actions of the males were again observed for thirty minutes.
Under these conditions the males collided sixteen times with the tube instead of ten. Moreover they seldom passed near the tube without some characteristic reaction. Usually they made one or two quick circles as they swam by, or even a somersault-like motion; these were observed fifteen times when the females were in the tube, never when they were not.
The experiment was now varied by dropping the small piece of tube to the bottom of the aquarium and recording the reactions of the males as before. During the half hour in which no females were in the tube, males collided with it thirteen times, but showed no inclination to play about it. After the introduction of the females eighteen collisions were observed and eight playing movements as males swam by. I am, therefore, thoroughly convinced that males respond to females even when they can not see them or come in contact with them. As the females are not known to have any power of producing sound, they probably give rise to some substance that serves as a scent for the males; in other words, the males are probably positively chemotropic toward the females.
Admitting this last conclusion, we have now all the facts needed for a satisfactory explanation of the daily migrations of Labidocerae. The females come to the surface as daylight vanishes because they are positively phototropic to weak light and because they are negatively geotropic. They descend from the surface into deeper water at the appearance of daylight because they are so strongly negatively phototropic to bright light that they overcome their negative geotropism. The males follow the females because they are probably positively chemotropic toward them.
THEORIES OF DAILY MIGRATIONS OF PELAGIC ANIMALS.
It is now comparatively simple to discuss the more important theories advanced to explain the daily migrations of pelagic animals. Chun's (1887)hypothesis that the migration depended primarily on temperature changes is absolutely without support from what has been learned about the daily movements of Labidocerae aestiva. Weismann's (1877) opinion that light governs the migration is very close to the truth, though it requires modification in the direction pointed out by Loeb (1894, p.67), who after having shown the general importance of light in the daily migrations of pelagic animals, expresses his belief "light is not the only physical influence
-------------------------------------------------------------------------
BULLETIN OF THE UNITED STATES FISH COMMISSION 1901.
Page 122
that determines the depth-migrations of sea animals." This has been abundantly demonstrated in Labidocera, where geotropism and chemotropism play parts as well as phototropism, and probably much the same complexity obtains in many other organisms whose depth-migrations, though made simultaneously, may be really the results of quite different causes.
CONCLUSIONS.
1. Labidocera aestiva has a specific gravity in reference to pure water of 1.109, to sea water 1.082.
2. It falls through sea water at the average rate of about half a centimeter a second (a fathom in six minutes). It swims upward at about one-third this rate.
3. In its fall it is oriented by the spread antennae, which keep the head uppermost.
4. Its locomotor movements are either rapid uniform runs, or leaps, or rarely some intermediate form of movement.
5. The runs are accomplished by the appendages between the anterior antennae and the tail.
6. The leaps can be carried out by the anterior antennae, by the tail, or by these combined, but not by the other appendages except in conjunction with either anterior antennae, or tail, or with both.
7. Labidocera aestiva may attach itself to fixed objects in the water by the anterior antennae. It may also hang from the surface of the water by these organs.
8. The anterior antennae perform at least three functions--mechanical orientation of the animal in falling, positive locomotion in leaping, and attachment to fixed objects or to the water film.
9. Males and females react in characteristically different ways to light, gravity, etc.
10. Females have a strong negative geotropism. Males have a weak negative geotropism.
11. The geotropism of Labidocera is not changed by shaking it in water or by other forms of mechanical stimulation. Increasing the density of the sea water from 1.025 to 1.035 and 1.050 increases slightly the negative geotropism of the males, but does not influence the females. Decreasing the density of the sea water from 1.025 to 1.020, 1.015, 1.010, 1.005, and 1.OO does not influence geotropism. Temperature changes between 10° and 35° C. have no influence on the geotropism of the males. The geotropism of the females is negative in cold water and positive in warm, the critical temperature being about 26° C.
12. Females have a strong positive phototropism for light of a low intensity. Males have a weak negative phototropism. The positive phototropism of the females is stronger than their negative geotropism. The phototropism of both sexes is unaltered by temperature changes between 10° and 35° C. It is also unaltered when the density of the sea water is varied from 1.050 to 1.060. Certain forms of mechanical stimulation (handling with a pipette, but not agitation in water) make females temporarily negatively phototropic and increase the natural negativity of males. Females are negative to light of high intensity (100-candle power incandescent light at 10 centimeters distance, and direct sunlight). Males are apparently indifferent to changes in light intensity.
-------------------------------------------------------------------
DAILY MIGRATIONS OF COPEPODS.
Page 123
13. Males are positive toward females and probably chemotropically positive.
14. Labidocera aestiva frequent the surface of the sea from sunset to sunrise. From sunrise to sunset it is presumably in deeper waters.
Its migrations are explained as follows: Females rise to the surface with the setting of the sun, because they are positively phototropic to faint light and negatively geotropic; they descend into deep water with the rising of the sun, because they are negatively phototropic to strong light (their negative geotropism being overcome by their negative phototropism); the males follow the female, in migration, because they are probably positively chemotropic toward the females.
Cambridge, Massachusetts, December 31, 1901.
PAPERS QUOTED.
Chun, C. 1887. Die pelagische Thierwelt in grosseren Meerestiefen und ihre Beziehungen zu der Oberflachenfauna. Bibliotheca Zoologica. Bd.1, pp. 1-66, Taf. I-V.
Famintzin, A. 1867. Die Wirkung des Lichtes auf Algen und einige andere ihnen nahe verwandte Organismen. Jahrbucher fur wissenschaftliche Botanik. Bd. vi, pp. 1-44.
Frandsen, P. 1901. Studies in the reactions of Limax maximus to directive stimuli. Proceedings American Acad. Arts and Sciences. Vol. xxxvii, pp. 185-227.
Fuchs, T. 1882. Was haben wir unter der "Tiefseefauna" zu verstehen und durch welches physikalische Moment wird das Auftreten derselben bedingt? Verhandlungen k. k. geologischen Reichsanstalt. Jahrgang 1882, pp. 55-68.
Holmes, S.J. 1901. Phototaxis in the Amphipoda. American Journ. of Physiology. Vol. v, pp. 211-234.
Giesbrecht, W. 1892. Systematik und Faunistik der pelagischen Copepoden des Golfes von Neapel. Fauna und Flora des Golfes von Neapel. XIX monographie. 831 p.54 Taf.
Groom, T. T., und Loeb, J. 1891. Der Heliotropismus der Nauplien von Balanus perforatus und die periodischen Tiefenwanderungen pelagischer Tiere.Biologisches Centralbatt. Bd. X, pp.160-177.
Loeb, J. 1890. Der Heliotropismuss der Thiere und seine Uebereinstimmung mit dem Heliotropismus der Pflanzen. Wurzburg. 118pp.
Loeb, J. 1893 Ueber kunstliche Umwandlung positiv heliotropischer Thiere in negativ helios-tropische und umgekehrt. Archiv Fir gesammte Physiologie. Bd. 54, pp. 81-107.
Loeb, J. 1894. On the influence of light on the periodical depth migration of pelagic animals. Bulletin U.S. Fish Commission for 1893, pp. 65-68.
Lubbock, J. 1884. On the sense of color among some of the lower animals. Part II. Journal of the Linnean Society. Zoology. Vol. XVIII, pp. 205-214.
MacBride W. E. 1899. The movement of copepods. Quart. Journ. Microscop. Science. Vol. XLII, pp. 505-507.
Peck, J. I. 1894. On the food of the menhaden. Bulletin U. S. Fish Commission for 1893, pp. 113-126, pls. 1-8.
___ 1896. The sources of marine food. Bulletin U. S. Fish Commission for 1895, pp. 351-368, pls. 68-71.
Radl, E. 1901. Ueber den Phototropismus einiger Arthropoden. Biologisches Centralblatt. Bd. XXI, p. 75-86.
Scourfield, D. J. 1900. Note on Scapholeberis mucronata and the surface-film of water. Journ. Quekett Microsc. Club. Vol. VII, pp. 309-312.
Towle, E. W. 1900. A study in the heliotropism of Cypridopsis. American Journ. of physiology. Vol. III, pp. 345-365.
Weisman, A. 1877. Das Thierleben im Bodensee. (Not accessible to me.)
Wheeler, W.M. 1900. The free-swimmimg copepods of the Woods Hole region. Bulletin U.S. Fish Commission for 1899, pp. 157-192.
Wilson, E.B. 1891. The heliotropism of Hydra. American Naturalist. Vol. XXV, pp. 413-433.
Yerkes, R. M. 1900. Reactions of Entomostraca to stimulation by light. II. Reactions of Daphnia and Cypris. American Journ. of Physiology. Vol. IV, pp. 405-422.
-----------------------------------------------------------------------
END